Berner Lab is pleased to share another publication featuring our trusted supplier Technobis and the Crystal16 platform.
Understanding Cocrystal Solubility
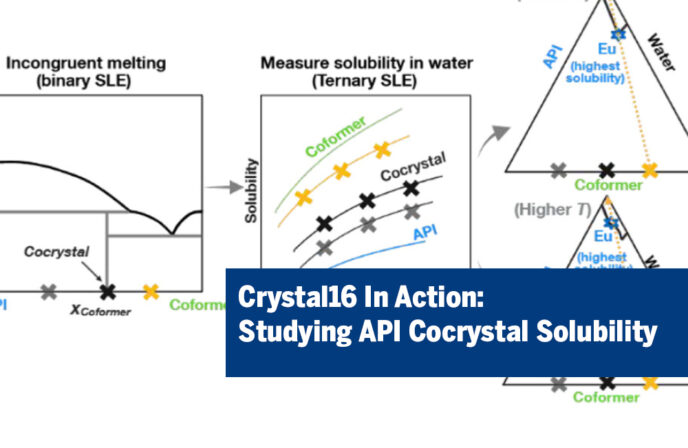
Pharmaceutical cocrystallization is a well-known approach for improving the solubility of poorly water-soluble APIs. However, predicting solubility becomes significantly more challenging when cocrystals melt incongruently.
A recent study, “Solubility of Incongruently Melting Active Pharmaceutical Ingredient Cocrystals: The Hydrochlorothiazide–Nicotinamide System“, investigated the aqueous solubility behavior of the 1:1 hydrochlorothiazide–nicotinamide (HCT–Nic) cocrystal using experimental measurements and thermodynamic modeling.
How Crystal16 Was Used
The Technobis Crystal16 was used to determine the aqueous solubility of hydrochlorothiazide (HCT), nicotinamide (Nic), and their binary mixtures across different compositions and temperatures. The experiments confirmed that solubility increased with temperature across all compositions, as expected. Nicotinamide provided modest solubility enhancement, with slightly greater improvement observed in Nic-rich mixtures and at higher temperatures.
Key Insights From The Study
The findings reveal important differences in cocrystal behavior depending on temperature. At 310.15 K, the system behaves as a simple eutectic with no cocrystal formation. At higher temperatures, however, a cocrystal phase appears and dissolves incongruently. Maximum HCT solubility occurs at the API-rich eutectic composition, with an enhancement factor of approximately 2.1–2.4. Notably, direct dissolution of the 1:1 cocrystal provides only limited solubility improvement.
We thank the authors for their valuable contributions and insights.
Read the full article here: https://pubs.acs.org/doi/10.1021/acs.molpharmaceut.5c01520?fig=fig4&ref=pdf
Want To Know More?
Contact Berner Lab and we will help you find the right Technobis solution for your solubility and crystallization research.

Jakob Meinhardt Nielsen